COVID-19 Rapid Antigen Testing

Please note: Due to TGA restrictions, we are unable to supply these products direct to consumers.
Since the beginning of the pandemic, Australia's COVID-19 response has relied on polymerase chain reaction testing (PCR tests) these are highly sensitive, but are expensive, and rely on transporting samples to a professional laboratory for analysis. As the Delta Variant began taking hold in 2021, we have seen more intrest in Rapid antigen testing, a solution that is faster, lower-cost, and can be provided at the point of care.
The value of rapid testing is that they can detect an active infection before symptoms become apparent, especially when used regularly as part of a testing regime, while the patient is shedding high levels of the virus and therefore has a high risk of transmitting it to others. However, it is critically important that those who do return a positive result from rapid antigen testing are immediately referred for PCR testing, as rapid antigen testing is more likely to provide false positives (and false negatives.)
Rapid antigen testing has been used extensively in other parts of the world such as the US and UK to complement PCR testing, and results are avaliable in as little as 15 minutes. Rapid antigen tests do not require complicated diagnostic equipment, making them perfect for use onsite at high-risk environments, or in regional and remote areas where a patient may need to travel some distance to reach a testing centre.
Can I purchase the test?
The tests can be supplied for use by specified health practitioners at the point of care to the following:
- Registered Medical Practitioners or Paramedics, or an organisation, business or institution that employs or engages a registered Medical Practitioner or Paramedic to perform or oversee performance of the test. The tests can only to be used to test employees or contractors of the organisation, business or institution, or a patient under the direct care of the Medical Practitioner or the Paramedic.
- Residential care (disability and rehabilitation facilities) and aged care facilities that employ or engage Health Practitioners (as defined by the Therapeutic Goods Act 1989) to conduct or perform the test. If the residential care or aged care facilities provide care in the home this condition would also allow for performance of the test to be conducted by a Health Practitioner or Paramedic. The tests can only be used to test residents, staff of, or visitors to, the residential care or aged care facility, or clients and staff of the home care service provider.
- Organisations, businesses, or institutions that employ or engage health practitioners or paramedics to conduct or oversee performance of the tests. For example, rapid antigen tests are being used in the mining sector consistent with these conditions. The tests can only be used to test staff or students of the organisation, business or institution, or a person who is a patient of a registered Dental Practitioner who requires an emergency dental procedure.
The tests can also be supplied to accredited laboratories and to Commonwealth, state or territory government departments, in cooperation with their relevant health departments.
Panbio Testing Procedure
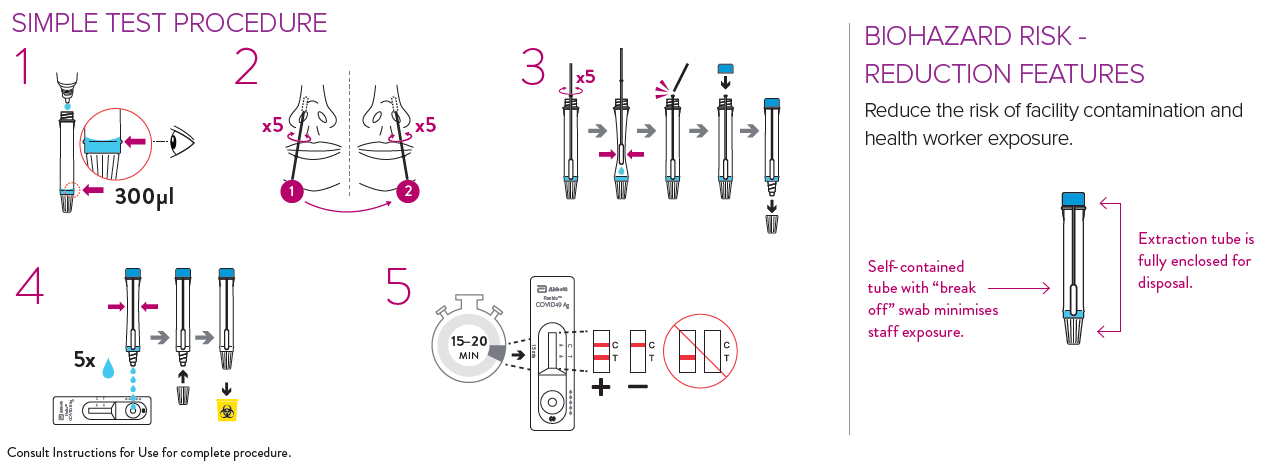
Panbio Training Videos
PanbioTM COVID-19 Ag Live Action Demo Video
Onsite Testing Procedure

OnSite training Video
What do I need to do to purchase the rapid antigen tests?
- Please make sure you watch the above training video before purchase and refer to it any time you require
- Complete the Customer Authorisation Form for the test you intend to purchase
- Wait for confirmation from Gildana Health Care that your account has been setup to purchase your Rapid Antigen Test.
- All purchases must be made on a Gildana Health Care 30-day account
- Our New Account Application form can be found here if required
- Call our Customer Service team on 03 9752 3455 or place your order online through our Web Self-Service System.
- After purchasing this product, you will need to attend webinar training provided by the manufacturer before you begin testing.
Be aware, we will need to pass your contact details onto the manufacturer so they can provide this training. - Further information on COVID-19 rapid antigen testing in general is avaliable from the TGA at the links below
Which products are available?
If you are after 1,000 or more tests please give us a call on 1300 556 917
| Product Code | Product Description | Test Type | UoM | Brand |
| 225139 | COVID-19 Rapid Antigen Test Panbio 25s | Nasopharyngeal | Box/25 | Abbott |
| 225167 | COVID-19 Rapid Antigent Test OnSite 20s | Nasopharyngeal | Box/20 | OnSite |
Supporting Documentation
General Information from the Therapeutic Goods Administration
Abbott Panbio Tests
- Panbio COVID-19 Antigen Test Brochure
- Panbio COVID-19 Antigen Test Instructions for Use
- Panbio COVID-19 Antigen Test Quick Reference Guide
- Abbott Panbio Rapid Antigen Test Customer Authorisation Form
OnSite Tests
 03 9752 3455
03 9752 3455